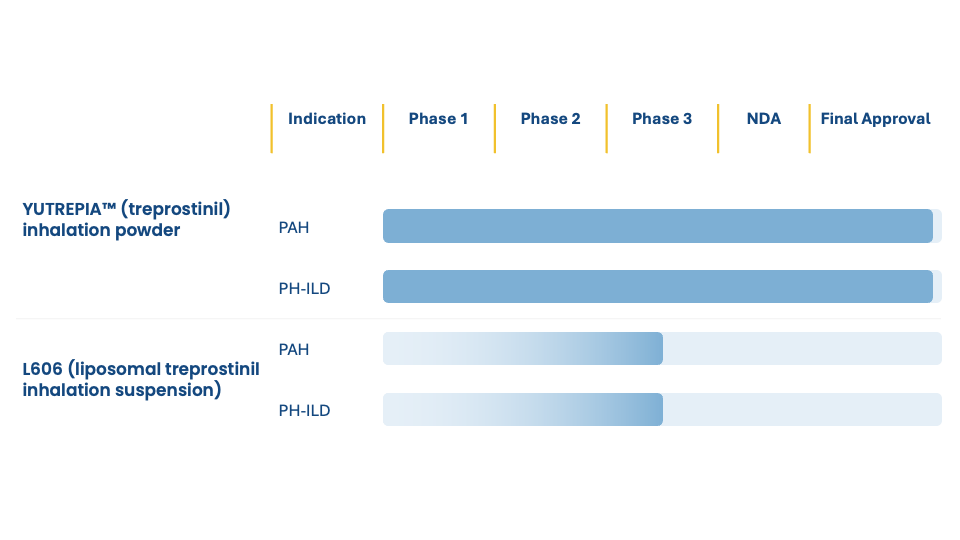
L606: A next-generation inhaled treprostinil
Developed in partnership with Pharmosa Biopharm, L606 (liposomal treprostinil inhalation suspension) is an investigational sustained-release liposomal formulation of treprostinil that has the potential to improve the delivery of treprostinil to the lung for patients living with PAH and PH-ILD.
Potential benefits include:
- Less frequent dosing (2x daily)
- Sustained drug exposure over 24 hours
- Improved tolerability with lower peak exposures and fewer systemic and inhalation-related issues
- Rapid nebulization with a hand-held portable system
L606 is delivered via rapid nebulization with a hand-held portable system and may offer therapeutic benefits for patients with PAH and PH-ILD.
L606 is composed of treprostinil encapsulated within liposomes (110 – 140 nm) in a sodium citrate-bicarbonate buffer. In this formulation, drug release is controlled by local levels of bicarbonate in the lung to extend apparent half-life.
Modeled plasma concentration following administration of TYVASO® or L6061
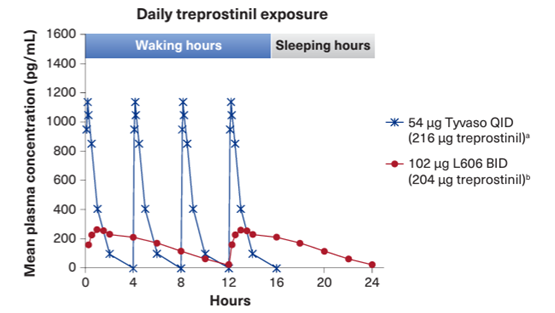
† Tyvaso data over 4 hours are from the comparative bioavailability study, and the remaining data are simulated to show Tyvaso 24-hour exposure.
‡ L606 data over 12 hours are from the comparative bioavailability study, and the remaining data are simulated to show L606 24-hour exposure.
TYVASO is a registered trademark of United Therapeutics Corporation.
Pioneers in rare cardiopulmonary disease
Explore the science behind our clinical trials.
References:
- Tully, J., Saggar, R., Prabel, J., Garcia, A., Patel, S., Chen, K., Kan, P. Clinical Pharmacokinetics of an Extended-Release Formulation of Inhaled Liposomal Treprostinil (L606) to Reduce Dosing Frequency. Poster presented at: Pulmonary Vascular Research Institute 2024 Annual Congress; February 2, 2024; London, England